Fabry Disease: Causes, Genetics, Symptoms, and Management
A comprehensive overview of Fabry disease, its genetic basis, signs and symptoms, inheritance patterns, and clinical implications.
Fabry Disease
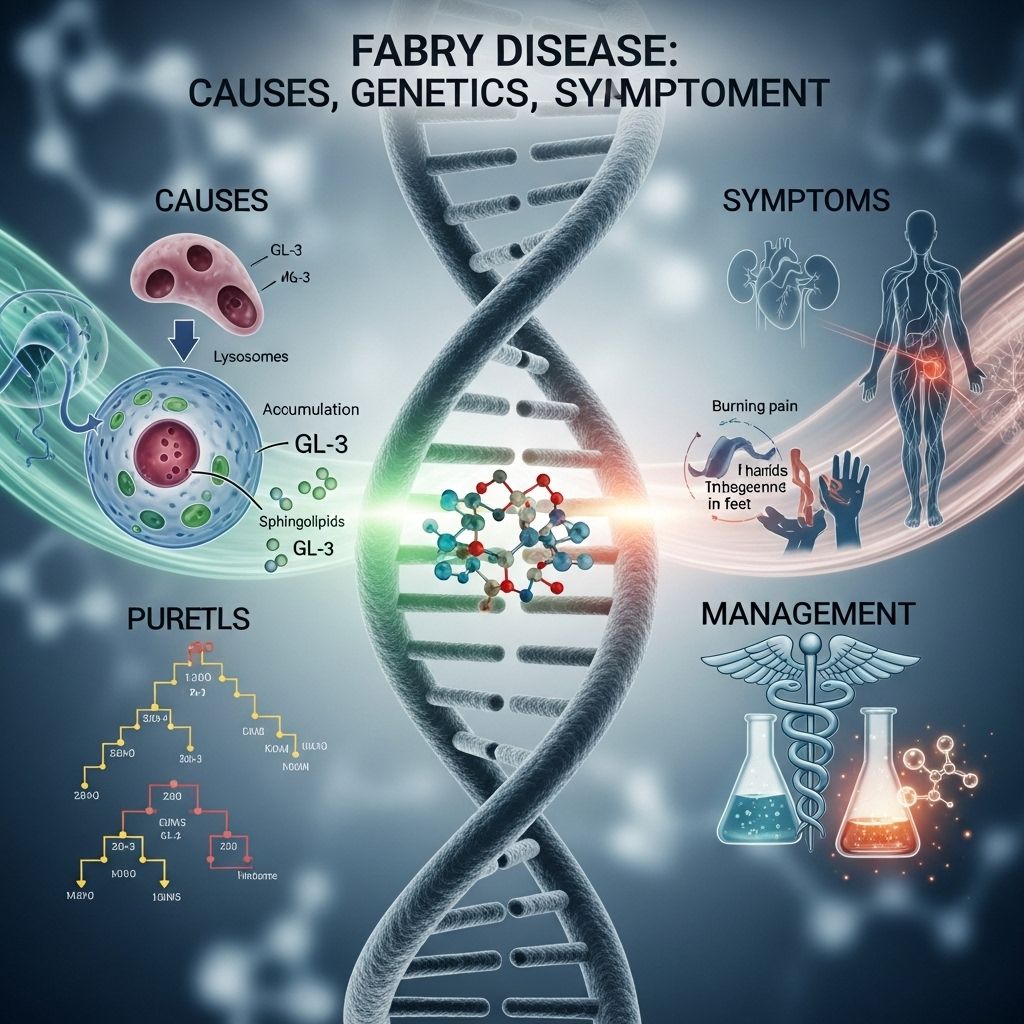
Fabry disease is an inherited metabolic disorder marked by the accumulation of a specific type of fat called globotriaosylceramide within various cells in the body. This buildup results from a deficiency in the enzyme alpha-galactosidase A. The disorder primarily affects blood vessels, skin, kidneys, heart, and the nervous system, causing a range of serious medical complications over time.
Overview
Fabry disease belongs to a group of conditions known as lysosomal storage disorders. Lysosomes act as the recycling centers within cells, breaking down unwanted substances. In Fabry disease, the failure to efficiently process particular fatty substances leads to their accumulation and subsequent organ damage.
- Globally rare: Fabry disease affects both males and females, though symptoms and severity may differ.
- X-linked inheritance: The responsible gene is located on the X chromosome.
- Disease onset and progression: It can manifest in childhood or adulthood depending on the type and severity.
Causes
Fabry disease is caused by variants (mutations) in the GLA gene.
- The GLA gene encodes the enzyme alpha-galactosidase A.
- This enzyme’s primary function is to break down globotriaosylceramide in lysosomes.
- Mutations alter the enzyme’s structure or production, resulting in limited or absent activity.
The consequences of these gene defects include:
- Disrupted fat processing: Inefficient breakdown of globotriaosylceramide results in its toxic accumulation.
- Widespread cellular impact: The stored fat primarily accumulates in blood vessels, kidney cells, heart muscle cells, and nervous system tissue.
- Progressive organ involvement: Ongoing buildup damages tissues, leading to a gradual onset of symptoms and complications.
Certain mutations in the GLA gene eliminate enzyme activity completely, causing the classic severe form of Fabry disease. Other variants allow for partial enzyme function, often leading to milder or later-onset forms that typically affect particular organs such as the heart, kidneys, or brain blood vessels.
Inheritance Pattern
Fabry disease is inherited in an X-linked pattern:
- X chromosome location: The GLA gene is found on the X chromosome, one of the two sex chromosomes.
- Affected males: Males have one X and one Y chromosome. Only one mutated copy of the gene (on the X chromosome) is required for males to develop the disease.
- Carrier and affected females: Females have two X chromosomes. One altered copy of the gene usually results in less severe or milder symptoms than in males, though some females may have no symptoms, while others experience significant medical issues.
- Inheritance risk: An affected male will pass the mutated gene to all daughters (making them carriers or affected), but not to sons. A carrier female has a 50% chance of passing the mutated gene to each child, regardless of sex.
Unlike most X-linked disorders, Fabry disease can result in marked health issues for females carrying only one defective gene. These women may show many typical symptoms of the disease, sometimes nearly as severe as those seen in males.
| Individual | X Chromosomes | Disease Impact |
|---|---|---|
| Male with mutated GLA | 1 X (mutated) / 1 Y | Typically severe, classic symptoms |
| Female with one mutated GLA | 1 X (mutated) / 1 X (normal) | Symptoms range from none to moderate or severe |
| Female with two normal GLA | 2 X (normal) | Unaffected |
Signs and Symptoms
The manifestations of Fabry disease are highly variable, depending on the degree of enzyme deficiency and the specific gene variant. Symptoms arise from progressive damage to blood vessels and tissues. They may begin in childhood or emerge later in adulthood.
- Pain and burning sensations: Most often in the hands and feet (acroparesthesias).
- Angiokeratomas: Distinctive tiny, dark red-to-purple skin lesions, typically around the groin, hips, and belly button.
- Corneal clouding: Opacities in the cornea detectable by eye exam (does not usually affect vision).
- Decreased sweat production: Many affected individuals cannot sweat normally.
- Gastrointestinal symptoms: Abdominal pain, diarrhea, nausea.
- Fatigue and weakness: Common and persistent.
- Progressive organ involvement:
- Kidneys: Development of proteinuria, gradual kidney dysfunction, eventual kidney failure.
- Heart: Thickening of the heart muscle (cardiomyopathy), arrhythmias, increased risk of heart attack and heart failure.
- Brain: Increased risk of stroke and other central nervous system issues.
Classic (Early-Onset) vs. Late-Onset Forms
| Form | Enzyme Activity | Typical Symptoms | Onset |
|---|---|---|---|
| Classic | Absent or near-absent | Early and multi-system symptoms | Childhood or adolescence |
| Late-Onset | Low, but not absent | Targeted organ involvement (e.g., heart) | Adulthood |
Clinical Impact and Complications
Over time, the progressive accumulation of globotriaosylceramide damages vital organs. Major complications include:
- Kidney failure requiring dialysis or transplantation
- Life-threatening heart disease
- Increased risk of high blood pressure and cardiac arrhythmias
- Early strokes or transient ischemic attacks
- Reduced life expectancy, typically due to cardiovascular or renal complications
Variability in Symptoms and Severity
Not all individuals with Fabry disease experience the same symptoms or degree of severity. This variability is largely due to:
- The specific GLA gene mutation involved
- Level of residual alpha-galactosidase A activity
- Random X-chromosome inactivation in females, leading to mosaic patterns of disease within tissues
- Other modifying genetic and environmental factors
Some females may be asymptomatic carriers, while others may have severe manifestations similar to affected males. Not all common features are present in every patient, and symptom onset can differ widely, even within a family.
Diagnosis
Fabry disease is identified through a combination of clinical evaluation, enzyme assays, and genetic testing:
- Clinical assessment: Based on characteristic symptoms (e.g., skin lesions, pain episodes, family history).
- Enzyme testing: Measures alpha-galactosidase A activity in blood; typically conclusive in males, may not always detect carrier females.
- Genetic analysis: Sequencing the GLA gene for definitive diagnosis and mutation identification.
- Family screening: Close relatives may be offered testing to determine risk or carrier status.
Management and Treatment
While there is currently no cure for Fabry disease, various therapies can help manage symptoms, slow disease progression, and reduce complications:
- Enzyme replacement therapy (ERT): Intravenous infusion of artificial alpha-galactosidase A to reduce substrate accumulation and potentially improve organ function.
- Chaperone therapy: Pharmacologic agents that stabilize and support function of residual enzyme in certain patients with amenable mutations.
- Symptomatic management:
- Pain relief (neuropathic pain medications such as anticonvulsants or antidepressants)
- Medication for heart and kidney complications (e.g., ACE inhibitors, angiotensin receptor blockers)
- Supportive therapies for gastrointestinal symptoms and fatigue
- Advanced organ support: Dialysis or kidney transplantation if renal failure develops; specialized cardiac care as needed.
- Regular monitoring: Ongoing evaluation by a multidisciplinary medical team.
Living with Fabry Disease
Living with Fabry disease requires vigilant medical care, lifestyle modifications, and emotional support. Many individuals benefit from:
- Comprehensive care coordination
- Pain management strategies
- Peer support groups and genetic counseling for affected families
- Early treatment initiation to maximize long-term outcomes
Frequently Asked Questions (FAQs)
Q: What is the main cause of Fabry disease?
A: Fabry disease is caused by mutations in the GLA gene, resulting in deficient or absent activity of the enzyme alpha-galactosidase A, leading to buildup of globotriaosylceramide in cells.
Q: Is Fabry disease inherited, and how?
A: Yes, Fabry disease follows an X-linked inheritance pattern. Males are generally more severely affected, while females may have variable symptoms or be asymptomatic carriers.
Q: What organs are most affected by Fabry disease?
A: The disorder primarily impacts the skin, nerves, kidneys, heart, and blood vessels, leading to pain episodes, organ dysfunction, and serious complications like renal failure or heart disease.
Q: Are there different types of Fabry disease?
A: Yes, the classic form (usually early onset with severe symptoms) and late-onset or variant forms (milder, often limited mainly to organs such as the heart or kidneys).
Q: Can Fabry disease be treated or cured?
A: While there is no cure, several therapies—including enzyme replacement therapy and chaperone therapy—can manage symptoms, reduce disease progression, and improve quality of life.
Q: What should families with a history of Fabry disease do?
A: Genetic counseling and family screening are recommended for relatives of individuals with Fabry disease to determine carrier status and identify those at risk early for timely intervention.
Additional Resources
- National Organization for Rare Disorders (NORD)
- Fabry Support & Information Group
- Genetic counseling services at major medical centers
Read full bio of medha deb